NO MORE WAITING
Fast, reliable, lab-grade diagnostic results at the point of care, for better outcomes.
Remove delays and guesswork. iaX delivers precision machine read & verified, quantitative results from a large range of rapid test brands at the point of care. Results securely synced to the cloud for immediate action.
PRECISION AT THE POINT OF NEED
From faint lines tolab-grade numbers
Machine-read, quantitative results remove subjectivity, reduce variability, and provide numerical data that you can trust for faster, more confident decisions.
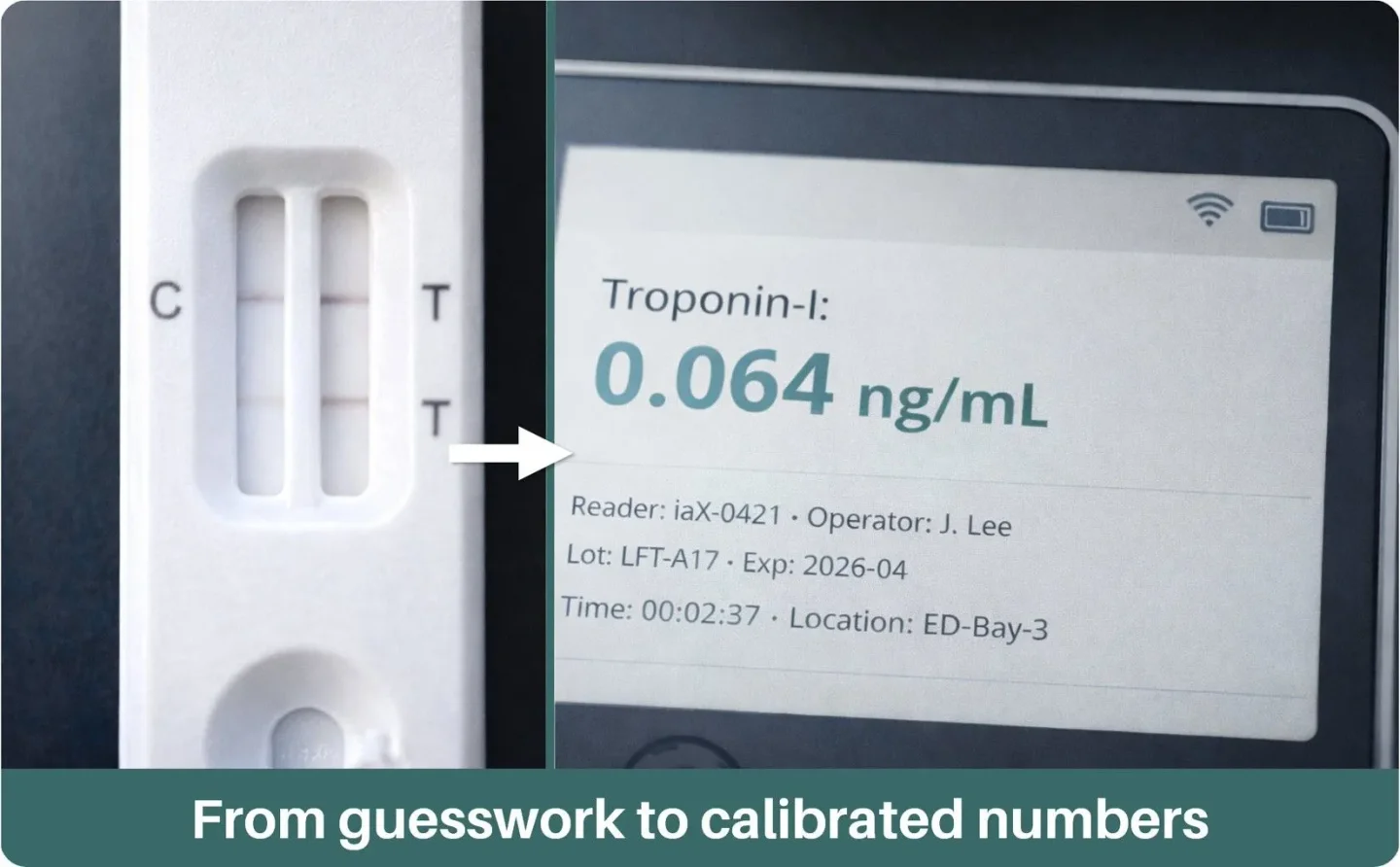

Small portable device, lab grade diagnostics.
The iaX is the world’s first smart, universal reader for rapid diagnostic tests.
It delivers accurate, connected results in seconds from multiple test manufacturers and securely uploads them to the cloud.
Compact and battery-powered, the iaX is for many sectors including human health, aquaculture, veterinary and more, bringing lab-grade diagnostics wherever they’re needed.

Rapid tests, reimagined with quantitative intelligence
Assaya supports a growing range of globally trusted rapid diagnostic tests and dip strips from leading manufacturers such as Abbott, BD, Roche, Abnova, and more, each qualified for use with the iaX platform.
Across human health, livestock, food safety, water, and environmental monitoring, iaX transforms standard rapid tests and dip strips into digitally verified, quantitative results you can act on immediately.
Instead of relying on subjective visual reads, iaX converts lines and color changes into precise, measurable data, removing human error and unlocking trend tracking, alerts, and cloud-based reporting. Every test becomes a trusted data point, fully traceable and ready for real-world decisions.
As new tests and strips are continuously qualified, the Assaya ecosystem keeps expanding, so you can use the diagnostics you already trust, with intelligence built in.

WE BRING THE LAB TO YOUR WORKPLACE
The iaX Intelligent Analyzerchanges the game.
Loaded with features, the iaX Intelligent Analyzer together with LFAs enable rapid, cost effective, lab-grade testing that transforms operational efficiency.
Optimize accuracy, reduce human error, and benefit from consistent precision reads across sites and teams.
Onsite results let you act now, not wait for the time-intensive lab loop.
Any brand of LFA can be qualified for use with iaX. No brand lock-in — one machine does it all.
Move beyond binary positive/negative outcomes with quantitative results and equivalent cycle threshold (eCT).
Auto-sync securely with full metadata for search, real-time reporting, dashboards, and compliance.
Small, portable, robust, battery-ready — ideal for clinics, farms, ships, factories, airports, and more.
Reduce manual records. Automation enables higher throughput for large-scale testing when it matters most.
Push results to EHR/LIMS/ERP and other systems when needed.
WHY IT MATTERS
Lateral flow assays are fast, but not flawless...iaX makes them accurate, connected and trusted.
Turn each test into a reliable, auditable data point you can act on, and stop letting ambiguity drive decisions.
Up to 80% of results are misread
Faint or borderline lines are highly prone to human error, with studies showing misreads can reach up to 80%, even among trained medical staff.
This most often results in false negatives, missed infections, and delayed action, increasing the risk of spread.
No real numbers,no clear action
Most rapid tests stop at positive/negative, without thresholds, severity levels, or trend context to guide decisions.
Without measurable data, you can't prioritize cases, track change over time, or intervene early, leaving critical actions to guesswork instead of evidence.
Quantitative results turn each test into a reliable data point that powers thresholds, trends, and clear triggers for action.
Same test.Different answers
No visibility across your operation
Results stay trapped at the test site, making it slow and difficult to detect patterns, outbreaks, or emerging risks.
Environmental factors distort results
Lighting, shadows, and background color can change how lines appear, skewing interpretation and confidence.
Scattered data, no traceability
Manual recording across paper, photos, and spreadsheets creates gaps, delays, and transcription errors, with no reliable audit trail.
WHO WE HELP
Innovative diagnostics solutions that work across industries and sectors
The iaX intelligent analyzer paired with the appropriate lateral flow assays allows rapid on-site lab-grade testing in human health, livestock, water and environment, and numerous other sectors.
Fast lab-grade diagnostics at the point of care.
On the scene diagnostics for first responders that save time and lives.
Rapid contaminant detection for safer water and ecosystems.
Detect disease early and protect your stock.
Field pathogen detection to protect yields.
On-farm diagnostics for herd and flock health.
Clinic and farm practice workflows.
Rapid pathogen and residue screening for compliance.
Solutions for government & NGOs. Emergencies and outbreaks.
HOW IT WORKS
From sample toresult in minutes
A simple streamlined workflow your team can run at the point of care.

Develop the assay according to its Instructions for Use (IFU).

Place the cartridge or strip into the iaX reader. Results are machine-read in seconds.

Full results including quantitative equivalents are stored securely with metadata for search and reporting.
HEAD TO HEAD COMPARISON
Lab-grade clarity,without the cost &the lab loop delay
Compare sensitivity, quantitation, speed, cost, field use, and data flow — and understand why a machine-read LFA wins on performance and practicality.
| Assay type | Sensitive | Quantitative | Cost | Speed |
|---|---|---|---|---|
| PCR | Yes | Yes | $$$ | No |
| ELISA | Yes | Yes | $$ | No |
| LFA | No | No | $ | Yes |
| LFA + iaX | Yes | Yes | $ | Yes |
| Assay type | Ease of use | In the field use | Ag & Ab | Direct to DB |
|---|---|---|---|---|
| PCR | No | No | No | No |
| ELISA | No | No | Yes | No |
| LFA | Yes | Yes | Yes | No |
| LFA + iaX | Yes | Yes | Yes | Yes |
| Assay type | Sensitive | Quantitative | Cost | Speed | Ease of use | In the field use | Ag & Ab | Direct to database |
|---|---|---|---|---|---|---|---|---|
| PCR | Yes | Yes | $$$ | No | No | No | No | No |
| ELISA | Yes | Yes | $$ | No | No | No | Yes | No |
| LFA | No | No | $ | Yes | Yes | Yes | Yes | No |
| LFA + iaX | Yes | Yes | $ | Yes | Yes | Yes | Yes | Yes |
What does Assaya do?
IN A NUTSHELL
iaX provides "super powers" to rapid tests.
Enabling precise, machine-read quantitative results, securely stored in the cloud, for cost-effective lab-grade diagnostics.
FREQUENTLY ASKED QUESTIONS
Answering your questions about LFAs and the Assaya iaX
Explore common questions and concerns to understand how this innovative technology streamlines testing.
LFAs combined with iaX smart readers deliver lab-grade accuracy with consistency and rapid results within minutes. While PCR tests are highly sensitive, LFAs provide faster turnaround on-site, enabling immediate decisions without sacrificing reliability.
The iaX reader standardizes test interpretation through machine-verified, quantitative reads. This eliminates subjective visual errors common in manual reading, ensuring consistent and objective results every time.
Yes, our iaX readers are Class I IVD devices designed for decentralized diagnostics and comply with applicable regulatory requirements, supporting adoption in clinical, environmental, agricultural, and public health settings.
Assaya offers cost-effective testing by reducing the need for centralized lab processing, minimizing sample transport, and speeding turnaround. Pricing varies by application and volume but typically offers significant savings compared to lab-based PCR testing.
Yes, iaX is universally compatible, supporting rapid tests from various manufacturers and across multiple sectors, allowing you to consolidate testing workflows on one easy-to-use platform. Not all LFAs have been onboarded and qualified for use with iaX, but we continue to add and support more.
After the sample is fully developed, and the LFA is ready to be read, the iaX can read and deliver the results in seconds, providing actionable insights in near real-time, a critical advantage in urgent diagnostic situations and fast-paced environments.
iaX automatically stores and timestamps all results in the cloud, enabling audit trails, real-time data sharing, and integration with EHR/LIMS/ERP systems through API and export options.
The solutions are designed for ease of use with minimal training; Assaya provides comprehensive support, onboarding, and resources to ensure smooth implementation and ongoing success.
iaX is small, robust, and hotspot-ready, built to operate reliably in the field, clinics, farms, ambulances, and other challenging environments.
Absolutely. Our technology supports diverse applications including food safety, environmental monitoring, public health, agricultural testing, and more, delivering fast, accurate insights where they are needed most.
BRING LAB-GRADE RESULTS TO YOUR POINT OF CARE
Start a conversation
Tell us about your workflow and where you need rapid, trusted results. We’ll walk you through device fit, assay options, and how data can integrate into your systems.



